Asked by Madyln Cunningham on Jul 31, 2024

Verified
Which of the following depictions most closely resembles the structure of the transition state for the following acid-base reaction? 
A) 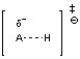
B) 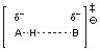
C) 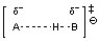
D) 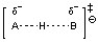
Transition State
A high-energy state during a chemical reaction, where reactants are transformed into products.
Acid-Base Reaction
A chemical reaction that involves the transfer of a hydrogen ion (H+) from an acid to a base.
- Acquire knowledge about the Hammond Postulate and its implications for predicting the courses and transition states of chemical reactions.

Verified Answer
ZK
Zybrea KnightAug 02, 2024
Final Answer :
C
Explanation :
The transition state for an acid-base reaction involves a partially formed and partially broken bond between the acid and base, as well as a partially formed and partially broken bond to the hydrogen or proton being transferred. Choice C best depicts this with the partially formed and partially broken bonds between H and O as well as between H and N. The other choices do not show the appropriate bond-breaking and bond-forming in the transition state.

Learning Objectives
- Acquire knowledge about the Hammond Postulate and its implications for predicting the courses and transition states of chemical reactions.
Related questions
Which of the Following Statements Is the Best Statement of ...
The Hydrogen Atom Abstraction Step in the Free Radical Chlorine ...
Which of the Following Correctly Expresses the Standard Gibbs Free ...
Predict the Major Organic Product of the Following Reaction Sequence ...
Provide the Major Organic Product Which Results When PhCHOHCH 3 Is ...