Asked by Ishaan Backliwal on May 07, 2024

Verified
Why does the diene shown below fail to undergo a Diels-Alder reaction with even the most reactive dienophiles? 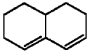
Diels-Alder Reaction
A chemical reaction between a conjugated diene and a substituted alkene, typically producing a cyclic compound.
Dienophiles
Chemical species that participate in Diels-Alder reactions by combining with dienes to form cyclic compounds.
- Recognize and explain the impact of molecular structure on reactivity and reaction mechanisms.

Verified Answer
ZK

Learning Objectives
- Recognize and explain the impact of molecular structure on reactivity and reaction mechanisms.
Related questions
Consider the Bond Dissociation Energies Listed Below in Kcal/mol ...
When O-Fluorotoluene Is Treated with Sodium Amide, the Product Is ...
Which of the Following Is the Most Polar Molecule: Methanol ...
What Type of Isomer Has the Same Four Atoms or ...
All Covalent Bonds Involve the Loss and Gain of Electrons