Asked by Gurwinder Bhullar on Jun 04, 2024

Verified
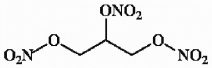
Nitroglycerine is formed by the reaction of glycerine (propane-1,2,3-triol) and nitric acid. Provide the structure of nitroglycerine.
Nitroglycerine
A highly volatile and explosive chemical compound used in dynamite and in medicinal preparations as a vasodilator.
Glycerine
A simple polyol compound used in foods, pharmaceuticals, and cosmetics for its moisturizing and solvent properties.
Nitric Acid
A highly corrosive and strong mineral acid with the formula HNO3, commonly used in the production of fertilizers, explosives, and in various nitration processes.
- Comprehend the fundamental principles of organic reactions and their processes.
- Distinguish among energy materials by examining their chemical configurations and responses.

Verified Answer

Learning Objectives
- Comprehend the fundamental principles of organic reactions and their processes.
- Distinguish among energy materials by examining their chemical configurations and responses.
Related questions
Predict the Major Product of the Following Dehydration Reaction
The Williamson Ether Synthesis Proceeds Via an ________ Mechanism ...
Given a Sample of (R)-2,3-Dimethylhexan-3-Ol, Which of the Following Would ...
Provide a Mechanism for the Reaction Shown Below
What Class of Organic Compound Is the Major Product of ...